|
11/3/2022 0 Comments Electron affinity This becomes especially important in biological systems, where molecules may have the same constituent atoms but the shape of the molecule is different – this is called a stereoisomer. 
An easy way to remember this is by knowing that the prefix ‘stereo’ means ‘3D’. 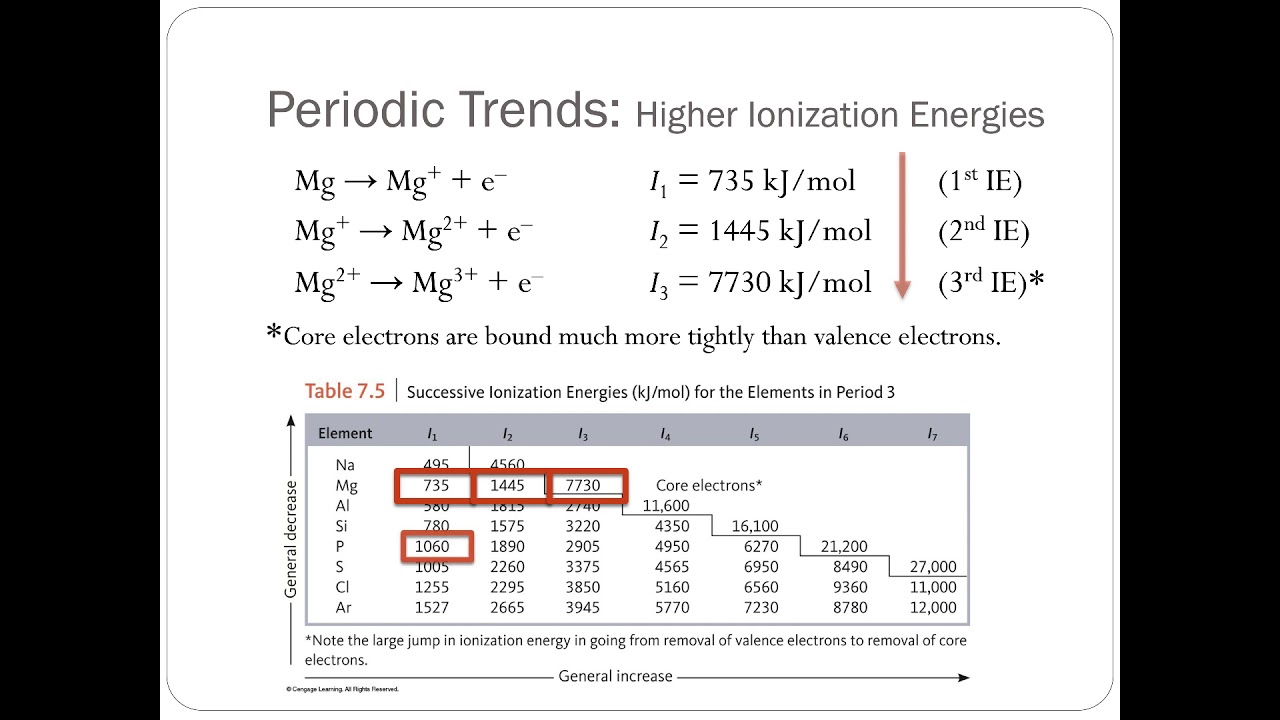
Stereochemistry relates to every aspect of the spatial relationships of atoms within a molecule. The affinity of a chemical species depends on the stereochemistry and the electrostatic interactions. For example, a positively charged molecule will have affinity for a negatively charged molecule. This is because the two species combine readily to form HF, as seen in this equation:Īffinity can also be referred to as the tendency of certain atoms (or molecules) to aggregate or bond together. For example, we say that fluorine has a very high affinity for hydrogen. There are multiple, more specific, uses of the term, such as chemical affinity and electron affinity.Ĭhemical affinity exclusively refers to the tendency of a chemical species to react with another. The term ‘ affinity’ is the tendency of a chemical species (such as an atom or a molecule) to react with another species to form a chemical compound. 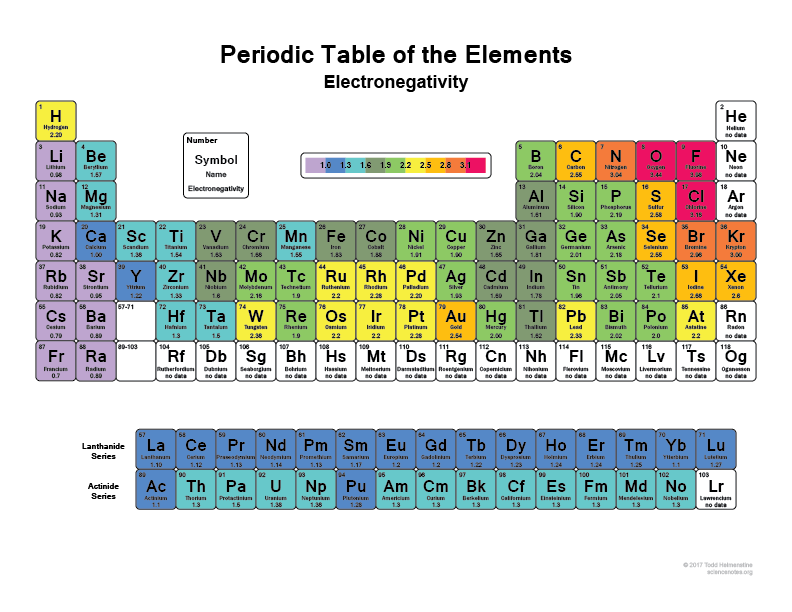
Drug affinity is how well a certain drug can bind to a receptor.Electron affinity is the amount of energy released or spent when an electron in added to a neutral atom.Chemical affinity depends on both stereochemistry and electrostatic interactions.Affinity is the tendency of a chemical species to react with another species to form a chemical compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
